Spike (B.1.1.529 BA.1, Omicron Variant) (SARS-CoV-2) Pseudotyped Lentivirus (Luc Reporter)
The pandemic coronavirus disease 2019 (COVID-19) is caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). As the first step of the viral replication, the virus attaches to the host cell surface before entering the cell. The viral Spike protein recognizes and attaches to the Angiotensin-Converting Enzyme 2 (ACE2) receptor found on the surface of type I and II pneumocytes, endothelial cells, and ciliated bronchial epithelial cells. Drugs targeting the interaction between the Spike protein of SARS-CoV-2 and ACE2 may offer protection against the viral infection. A variant called B.1.1.529 BA.1 (also known as the Omicron Variant) was identified in South Africa in November of 2021. This variant has a large number of mutations that allow the virus to spread more easily and quickly than other variants.
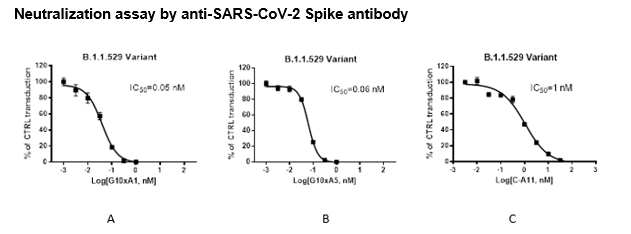
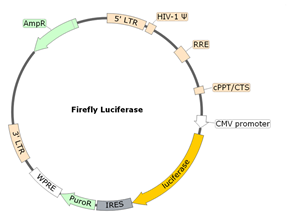
The Spike (B.1.1.529 Variant) (SARS-CoV-2) Pseudotyped Lentivirus were produced with SARS-CoV-2 B.1.1.529 BA.1 Variant Spike (Genbank Accession #QHD43416.1 with B.1.1.529 BA.1 mutations; see below for details) as the envelope glycoproteins instead of the commonly used VSV-G. These pseudovirions contain the firefly luciferase gene driven by a CMV promoter (Figure 1), therefore, the spike-mediated cell entry can be measured via luciferase activity. The Spike (B.1.1.529 Variant) (SARS-CoV-2) pseudotyped lentivirus can be used to measure the activity of neutralizing antibody against SARS-CoV-2 B.1.1.529 BA.1 variant in a Biosafety Level 2 facility.
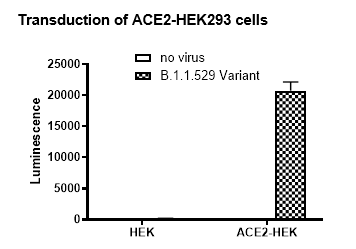
As shown in Figures 2 and 3, the Spike Omicron pseudovirus has been validated for use with target cells ACE2-HEK293 (which overexpress ACE2; BPS Bioscience, #79951).
Spike Mutations in B.1.1.529 BA.1 Variant:
A67V, Δ69-70, T95I, G142D, Δ143-145, Δ211, L212I, ins214EPE, G339D, S371L, S373P, S375F, K417N, N440K, G446S, S477N, T478K, E484A, Q493R, G496S, Q498R, N501Y, Y505H, T547K, D614G, H655Y, N679K, P681H, N764K, D796Y, N856K, Q954H, N969K, L981F
| Name | Ordering Information |
| Thaw Medium 1 or HEK293 Growth Medium | BPS Bioscience #60187 |
| ACE2-HEK293 Recombinant Cell Line | BPS Bioscience #79951 |
| Spike Neutralizing Antibody (Clone C-A11) (SARS-CoV-2) | BPS Bioscience #101024 |
| Spike Neutralizing Antibody (Clone G10xA1) (SARS-CoV-2) | BPS Bioscience #101326 |
| Spike Neutralizing Antibody (Clone G10xA5) (SARS-CoV-2) | BPS Bioscience #101327 |
| 96-well tissue culture treated, white clear-bottom assay plate | Corning #3610 |
| ONE-Step™ luciferase assay system | BPS Bioscience #60690 |
The lentiviruses were produced from HEK293T cells. Supplied in medium containing 90% DMEM + 10% FBS.
Media Formulation
Thaw Medium 1 (BPS Bioscience #60187):
MEM medium supplemented with 10% FBS, 1% non-essential amino acids, 1 mM Na pyruvate, 1% Penicillin/Streptomycin.