Bispecific Antibodies & BiTE® (Bispecific T-Cell Engager) Services
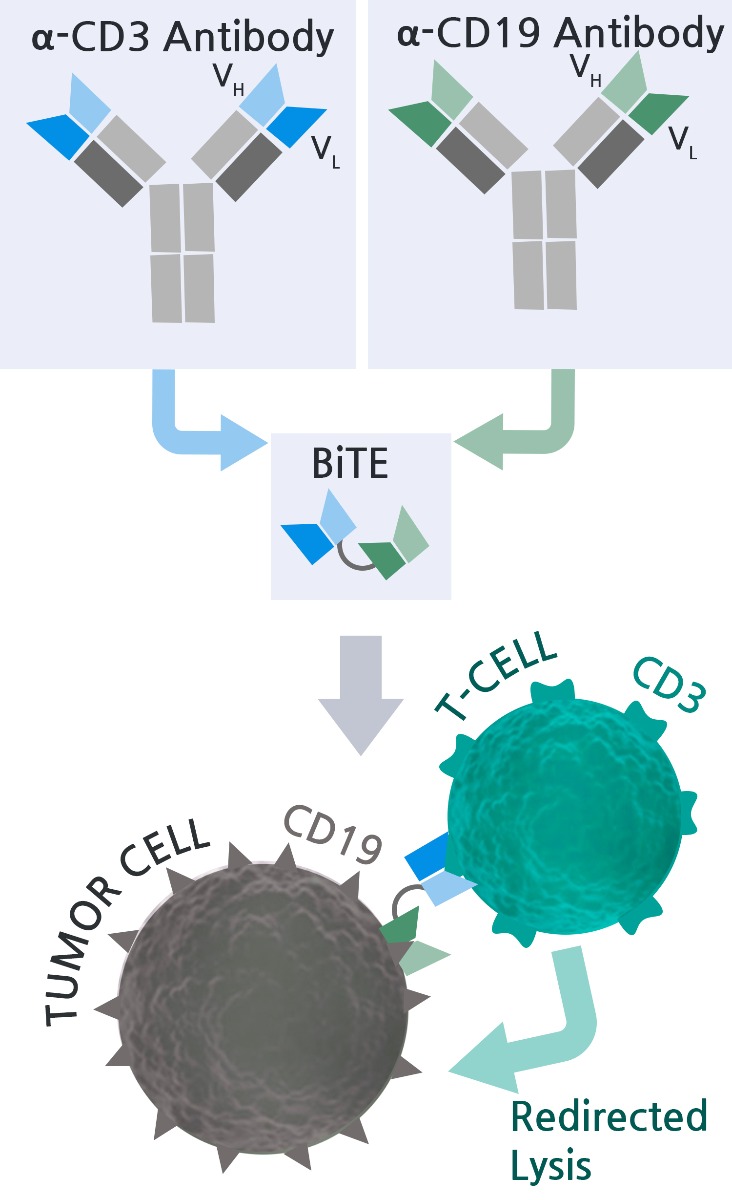
CD3/CD19 BiTE® - Mechanism of Action
The CD3/CD19 BiTE® is a biopharmaceutical drug known as a Bi-specific T-cell engager for a second-line treatment for Philadelphia chromosome-negative relapsed or refractory acute lymphoblastic leukemia. It binds to CD19 on cancer cells (e.g. Raji cells) and CD3 on T cells simultaneously, thus bringing T lymphocytes closer to cancer cells expressing specific cancer antigens (e.g. CD19). The binding event potentiates unstimulated T-cells and induces direct cytotoxicity against CD19+ cancer cells.
BPS has demonstrated the capability of assessing the functional activities of a commercially available CD3/CD19 BiTE® using our reporter cell lines, Jurkat/NFAT-luc or Jurkat/IL2-Luc.
Experimental Procedures
First, Jurkat effector cells with endogenous TCR/CD3 and transfected reporter NFAT-Luc or IL-2 promoter-Luc cells are incubated with increasing concentrations of the CD3/CD19 BiTE®in the presence or absence of CD19+ Raji cells. Secondly, the CD3/CD19 BiTE® simultaneously binds to TCR/CD3 on the Jurkat reporter cells and tumor antigen CD19 on the target Raji cells. Finally, its binding stimulates NFAT or IL-2 luciferase activity and the luciferase activity is assessed by our ONE-Step™ Luciferase Assay System.

BsAb-related Services
BPS offers customized services for generating BiTE® constructs and producing BiTE® antibodies, measuring the affinity for BiTE® antibody binding to antigen using interferometry (Gator™, Probe Life) or ELISA-based assay, and assessing T cell activation using reporter cell-based assays. BPS also offers over 200 cell lines expressing antigens on cancer cells on CHO or HEK293 cells, including our CD19-CHO Cell Line and several reporter cell lines including NFAT(Luc)/Jurkat cell lines for BiTE® construct evaluation in a reporter cell-based assay.
If you have further questions or would like to request a quote, please feel free to contact us.
References
1. Labrijn, A.F., Janmaat, M.L., Reichert, J.M., and Parren, P.W.H. (2019) Bispecific antibodies: a mechanistic review of the pipeline, Nature Reviews Drug Discovery 19, 585-608
2. Sedykh, S.E., Prinz, V.V., Buneva, V.N., and Nevinsly, G.A. (2018) Bispecific antibodies: design, therapy, perspectives, Drug Design, Development and Therapy 12, 195-208
Quote Request
Related Services
Biochemical Assay Development
Cell-Based Assay Development
CAR T-Cell Development
Protein Expression
Custom Cell Line Development
Screening & Profiling
Related Products
Anti-CD19-Anti-CD3 Bispecific Antibody
NFAT Reporter (Luc) – Jurkat Recombinant Cell Line
IL-2-Luciferase Reporter (Luc) - Jurkat Cell Line
CD19, Fc-Fusion (IgG1), Avi-Tag, Biotin-Labeled
ONE-Step™ Luciferase Assay System