Anti-BCMA CAR Lentivirus (Clone C11D5.3 ScFv-CD8-4-1BB-CD3ζ)
The anti-BCMA CAR lentiviruses are replication incompetent, HIV-based, VSV-G-pseudotyped lentiviral particles that are ready to infect almost all types of mammalian cells, including primary and non-dividing cells. These viruses transduce the ScFv (single-chain variable fragment) of anti-BCMA (clone C11D5.3) linked to a 2nd generation CAR (Chimeric Antigen Receptor) containing CD8 hinge and transmembrane domains, and the 4-1BB and CD3ζ signaling domains (Figure 1). The lentiviruses also transduce a puromycin selection gene.
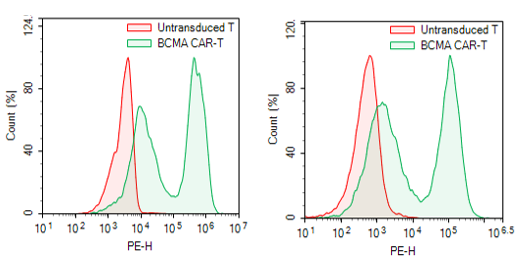
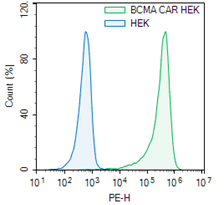
The mouse anti-BCMA antibody portion of this CAR construct recognizes human BCMA.
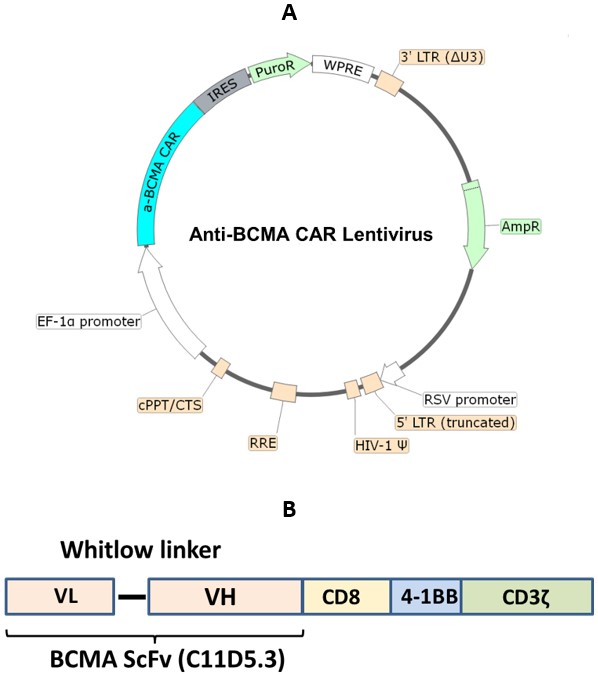
Figure 1. (A) Schematic of the lenti-vector used to generate the anti-BCMA CAR lentivirus with puromycin selection, and (B) Construct diagram showing components of the anti-BCMA CAR
| Name | Ordering Information |
| PBMC, Frozen | BPS Bioscience #79059 |
| Human Interleukin-2 | BPS Bioscience #90184 |
| EasySep™ Human CD4+ T Cell Isolation Kit | Stemcell Technologies #17952 |
| EasySep™ Human CD8+ T Cell Isolation Kit | Stemcell Technologies #17953 |
| Human CD3/CD28/CD2 T Cell Activator | Stemcell Technologies #10970 |
| BCMA, Fc-fusion (IgG1), Avi-Tag, Biotin-Labeled | BPS Bioscience #79467 |
| PE-Streptavidin | Biolegend #405203 |
| BCMA / Firefly Luciferase - CHO Recombinant Cell Line | BPS Bioscience #79724 |
| Firefly Luciferase - CHO Recombinant Cell Line | BPS Bioscience #79725 |
| Firefly Luciferase-RPMI 8226 Recombinant Cell Line | BPS Bioscience #79834 |
| ONE-Step™ Luciferase Assay System | BPS Bioscience #60690 |
The lentiviruses were produced from HEK293T cells, concentrated and resuspended in DMEM.
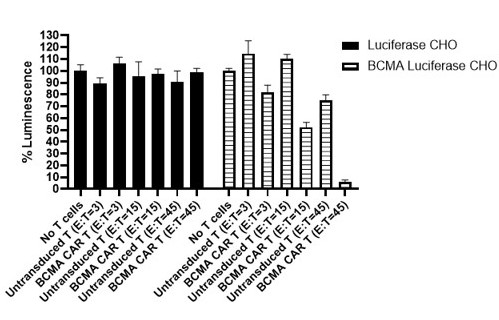
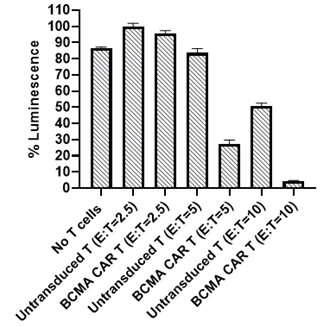
B-cell maturation antigen (BCMA), also known as CD269 or tumor necrosis factor receptor superfamily member 17 (TNFRSF17) is a cell surface receptor of the TNF receptor superfamily that recognizes B-cell activating factor (BAFF). BCMA is preferentially expressed in mature B lymphocytes and on Multiple Myeloma (MM) cells. BCMA is a highly attractive target antigen for immunotherapy because of its restricted expression in nonmalignant tissue but almost universal expression on MM cells. To date, the FDA has approved two BCMA CAR-T therapies for the treatment of multiple myeloma.