Dual Epitope Anti-BCMA CAR-T Cells
The Dual Epitope Anti-BCMA CAR-T Cells are generated via high-titer lentiviral transduction of human primary CD4+ and CD8+ T cells with the SIN Anti-BCMA CAR Lentivirus (VHH1/VHH2 ScFv-CD8-4-1BB-CD3ζ) (#78783). These ready-to use CAR (chimeric antigen receptor)-T cells express an anti-BCMA CAR consisting of a ScFv (single-chain variable fragment) that recognizes two BCMA epitopes (VHH1 and VHH2) linked to a CD8 hinge and transmembrane domains, and the 4-1BB and CD3ζ signaling domains (Figure 1).
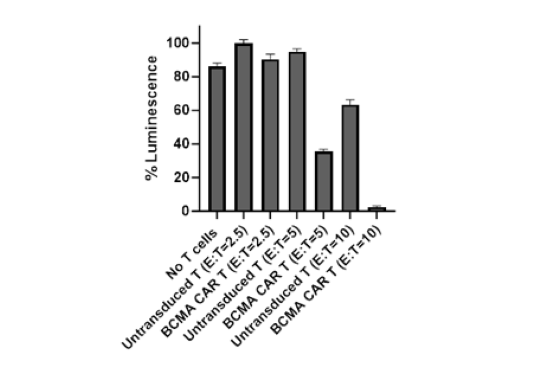
These CAR-T cells have been validated by flow cytometry (to determine the CAR expression) and in co-culture cytotoxicity assays.

Figure 1: Construct diagram showing components of the anti-BCMA CAR expressed in Dual Epitope Anti-BCMA CAR-T Cells.
| Name | Ordering Information |
| Human Interleukin-2 Recombinant | BPS Bioscience #90184 |
| Human CD3/CD28/CD2 T Cell Activator | Stemcell Technologies #10970 |
| BCMA, Fc-Fusion, Avi-Tag, PE-Labeled Recombinant | BPS Bioscience #100733 |
| Untransduced T Cells (as Negative Control for CAR-T cells) | BPS Bioscience #78170 |
| Firefly Luciferase CHO Cell Line | BPS Bioscience #79725 |
| BCMA/Firefly Luciferase CHO Cell Line | BPS Bioscience #79724 |
| Firefly Luciferase RPMI8226 Cell Line | BPS Bioscience #79834 |
| Thaw Medium 3 | BPS Bioscience #60186 |
| Thaw Medium 10 | BPS Bioscience #79704 |
| ONE-Step™ Luciferase Assay System | BPS Bioscience #60690 |
| Luminometer |
Recommended Anti-BCMA CAR-T Cell Medium: TCellM™ (#78753) supplemented with 10 ng/ml Interleukin-2 (#90184).
The cells have been screened to confirm the absence of Mycoplasma species.
BCMA (B-cell maturation antigen), also known as CD269 or tumor necrosis factor receptor superfamily member 17 (TNFRSF17), is a cell surface receptor of the TNF (tumor necrosis factor) receptor superfamily that recognizes BAFF (B-cell activating factor). BCMA is preferentially expressed in mature B lymphocytes and in Multiple Myeloma (MM) cells. BCMA is a highly attractive target antigen for immunotherapy because of its restricted expression in nonmalignant tissue but almost universal expression on MM cells. BCMA CAR-Ts are primary T cells engineered by lentiviral transduction to express a fully human-specific BCMA-CAR. CAR-T cells targeting BCMA have shown clinical anti-MM activity, and in 2022, the FDA granted authorization to use LCAR-B38M CAR-T Cell immunotherapy in MM. Active research into the development of BCMA targeting CARs for the treatment of several oncogenic disorders is ongoing, and represents a promising therapeutic avenue in cancer therapy.
Ghosh A., et al., 2017 Leuk Lymphoma. 6:1-12
Sanchez E., et al., 2018 Expert Rev Mol Diagn. 7:1-11.
Sohail A., et al., 2018 Immunotherapy. 10(4):265-282.
Sidaway P., et al., 2016 Nat Rev Clin Oncol. 13(9):530.