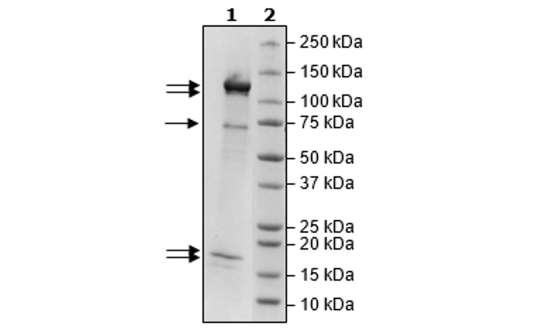
Rbx1/CUL4B/DDB1/DCAF15/DDA1 Complex Recombinant
Recombinant complex of human RBx1 (ring-box 1), CUL4B (cullin 4B), DBB1 (damage specific DNA binding protein 1), DCAF15 (DDB1 and CUL4 associated factor 15), and DDA1 (DET1 and DDB1 associated 1). RBx1, full length, encompassing amino acids 2-108(end). This construct contains an N-terminal His-tag (6xHis). CUL4B, encompassing amino acids 3-913(end). This construct contains an N-terminal His-tag (6xHis). DBB1, full length, encompassing amino acids 2-1140(end). This construct contains an N-terminal FLAG-tag. DCAF15, full length, encompassing amino acids 2-600(end). This construct contains an N-terminal FLAG-tag. DDA1, full length, encompassing amino acids 2-102(end). This construct contains an N-terminal FLAG-tag. These recombinant proteins are co-expressed in a HEK293 expression system and affinity purified.
≥90%
Aqueous buffer solution
40 mM Tris-HCl, pH 8.0, 110 mM NaCl, 2.2 mM KCl, 0.04% Tween-20, 20% glycerol
Covalent conjugation to ubiquitin (Ub) is a major post-translational modification regulating protein stability, function, and localization. Ubiquitination is the concerted action of three enzymes: a Ub-activating enzyme (E1), a Ub-conjugating enzyme (E2), and a Ub ligase (E3), which directs the last step of the Ub-conjugating cascade by binding to both an E2∼Ub conjugate and a substrate protein. This step ensures the transfer of Ub from E2∼Ub to the substrate, leading to its mono- or poly-ubiquitination.
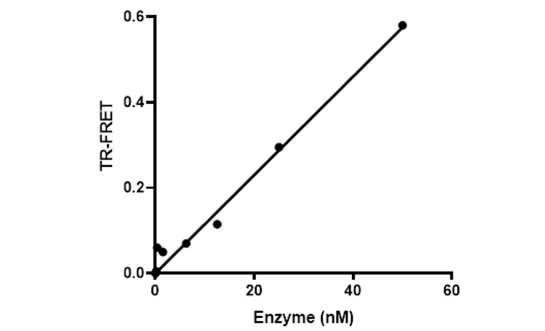
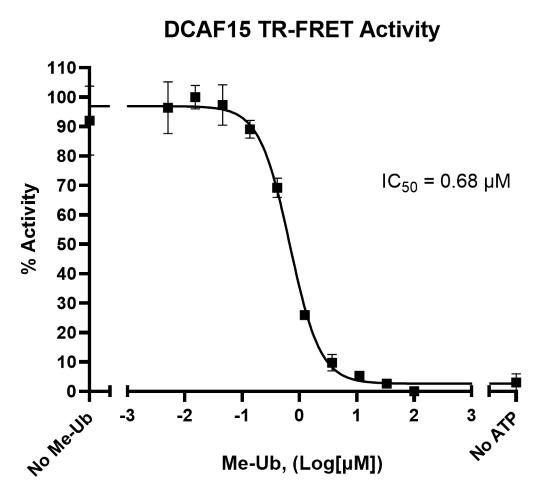
DCAF15 (DDB1 and CUL4-associated factor 15) associates with CUL4A or CUL4B, Rbx1, DDB1 and DDA1 to confer E3 ligase activity to the complex. DCAF15 interacts with and ubiquitinates the major transcription factor ZEB1, which is involved in the activation of the epithelial-mesenchymal transition (EMT) in metastatic cancer cells. Furthermore, DCAF15 is necessary for ubiquitination of the RNA splicing factor RBM39, the degradation of which leads to enhanced sensitivity of hematopoietic and lymphoid cancer cells to drug-induced cytotoxicity. Therefore, DCAF15 is an attractive potential drug target for cancer therapy. Like most E3 ligases, DCAF15 ubiquitinates itself.