C-CBL-Driven SRC Ubiquitination Intrachain TR-FRET Assay Kit
The C-CBL-driven SRC Ubiquitination Intrachain TR-FRET Assay Kit utilizes a Europium cryptate-labeled Ubiquitin Donor and a Cy5-labeled Ubiquitin Acceptor to complete the TR-FRET pairing. Since both the TR-FRET donor and acceptor are incorporated into poly-ubiquitin chains, this assay does not detect mono-ubiquitination. The FRET-based format requires no time-consuming washing steps, making it especially suitable for HTS applications as well as real-time analyses of polyubiquitination.
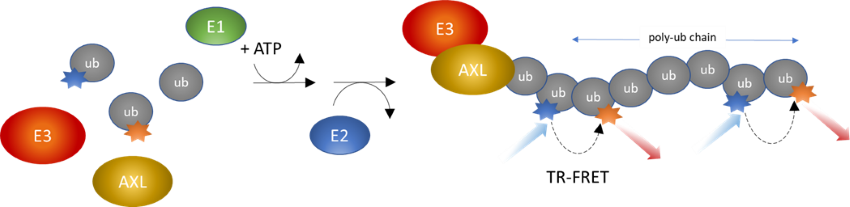
Figure 1: C-CBL-driven SRC ubiquitination intrachain TR-FRET Assay Kit schematic.
Need us to run inhibitor screens or profile your compounds against C-CBL-Driven SRC Ubiquitination? Check out our Ubiquitination Screening Services.
- Fluorescent microplate reader capable of measuring Time Resolved Fluorescence Resonance Energy Transfer (TR-FRET)
- Adjustable micropipettor and sterile tips
- Rotating or rocker platform
| Catalog # | Name | Amount | Storage |
| 80301 | UBE1 (E1)* | 40 µg | -80°C |
| 80314 | UBCH5b (E2)* | 60 µg | -80°C |
| 100370 | C-CBL, GST-Tag* | 8 µg | -80°C |
| 40484 | SRC, His-Tag* | 12 µg | -80°C |
| 78307 | TRF Ubiquitin Mix (200x) | 40 µl | -80°C |
| ATP (4 mM) | 2 x 1 ml | -80°C | |
| U2 Assay Buffer | 2 x 10 ml | -80°C | |
| 79969 | White, nonbinding, low volume 384-well microtiter plate | Room Temp |
*The initial concentration of enzyme is lot-specific and will be indicated on the tube containing the protein.
Covalent conjugation to ubiquitin (Ub) regulates protein stability, function, and localization. Ubiquitination is the concerted action of three enzymes: a Ub-activating enzyme E1, a Ub-conjugating enzyme E2, and a Ub ligase E3. The specificity and efficiency of ubiquitination are largely determined by the E3 enzyme, which directs the last step of the Ub-conjugating cascade by binding to both an E2∼Ub conjugate and a substrate protein.
Casitas B-lineage lymphoma (C-CBL) is the RING-type E3 ligase that that functions as a negative regulator of T cell activation. It contains an N-terminal tyrosine kinase binding domain, a SRC homology domain, and the RING domain responsible for its catalytic function. Additionally, C-CBL contains proline-rich regions mediating the association with various targets, as well as a ubiquitin-associated (UBA) domain for ubiquitin binding and dimerization. C-CBL interacts with phosphorylated proteins. C-CBL interacts with a large number of target proteins implicated in the control of cell proliferation and differentiation. The ubiquitin ligase activity of C-CBL is regulated by the phosphorylation of Tyrosine 371, which C-CBL from its auto-inhibitory conformation, allowing binding to E2 and substrates. Kinases that phosphorylate C-CBL, such as AXL, Tyro3, and SRC, also serve as substrates for ubiquitination.