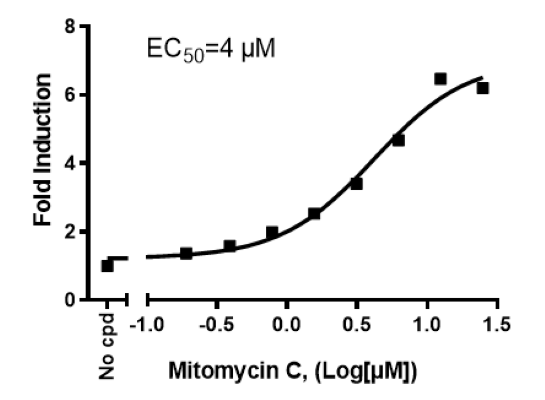
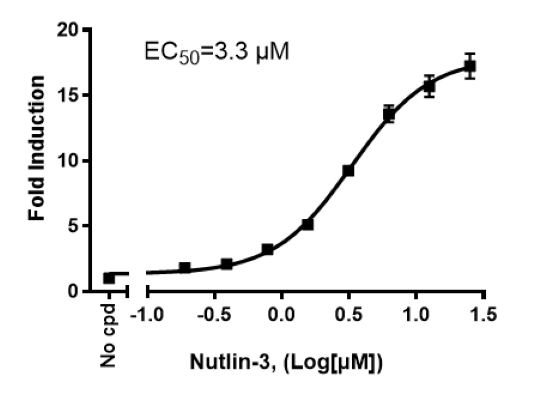
p53 Luciferase Reporter HCT116 Cell Line
The p53 Luciferase Reporter HCT116 Cell Line is an HCT116 cell line (expresses wild-type p53) that expresses Firefly luciferase under the control of the p53 response element. This cell line monitors the activity of the p53 signaling pathway. This cell line has been validated in activation assays in response to Mitomycin C, Nutlin-3, and Doxorubicin.
Interested in screening and profiling inhibitors or activators of the p53 pathway without the need to purchase and license the cell line? Check out our Cell Signaling Pathway Screening.
Purchase of this cell line is for research purposes only; commercial use requires a separate license. View the full terms and conditions.
Media Required for Cell Culture
| Name | Ordering Information |
| Thaw Medium 7 | BPS Bioscience #60185 |
| Growth Medium 7C | BPS Bioscience #78076 |
Materials Used in Cellular Assay
| Name | Ordering Information |
| Assay Medium 7A | BPS Bioscience #78673 |
| Mitomycin C | BPS Bioscience #27763 |
| Nutlin-3 | BPS Bioscience #27711 |
| Doxorubicin | Sigma #D1515 |
| Clear-bottom, white 96-well tissue culture-treated plate | Corning #3610 |
| ONE-Step™ Luciferase Assay System | BPS Bioscience #60690 |
| Luminometer |
The cell line has been screened to confirm the absence of Mycoplasma species.
p53 is a transcription factor and tumor suppressor very frequently mutated in human cancer and often termed “guardian of the genome”. Activated by DNA damage, oxidative stress, or deregulated oncogene expression, p53 binds to a specific site in the promoter region of target genes and leads to the transcriptional activation of downstream genes involved in DNA repair, cell cycle arrest, senescence, and apoptosis. p53 levels are normally low, as the protein is targeted for degradation by MDM2 (mouse double minute 2). Upon activation by oxidative stress, or other cellular stress stimuli, p53 levels increase in the cells and the protein is activated. Inactivation of p53 promotes genome instability and directly contributes to cell transformation, and mutations in p53 contribute to 50% of the tumors. Using p53 as a therapeutic target requires regulation of p53 function to endogenous levels, since high levels of p53 seem to result in premature aging. p53 itself is regulated by multiple molecular mechanisms. An understanding of the pathways that regulate p53 is critical for the development of therapeutic modalities that restore its function and levels to endogenous levels.
Hafner A., et al., 2019 Nature Reviews Molecular Cell Biology 20:199-210.