HVEM, LIGHT & BTLA: New Immunotherapy Target
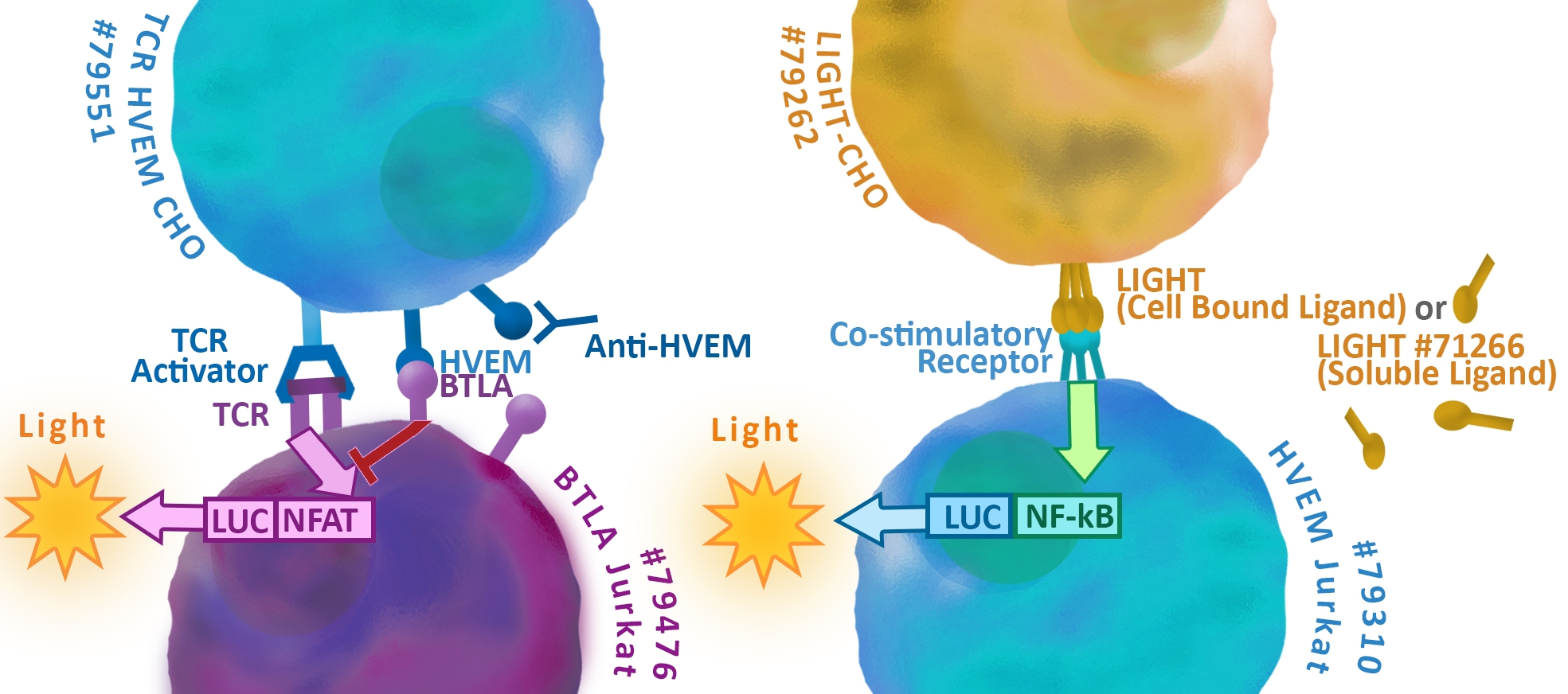
The checkpoint receptors HVEM, LIGHT, CD160, and BTLA are part of a complex network of overlapping receptor interactions that function in both immune stimulation and suppression1, 2 . This regulatory function has made them therapeutic targets for treatment of cancer, autoimmune diseases and allergies, and for improved methods of organ transplants. While presenting opportunities for therapeutic development, this system creates challenges because potential therapies may impact interactions beyond their intended targets resulting in unintended consequences.
HVEM is a molecular switch that acts both as an immune system stimulator3, 4 and inhibitor5,6,7. HVEM is expressed in a variety of immune cells including T cells, B cells, natural killer cells, dendritic cells and endothelial cells8. It is also implicated in immune evasion of tumors through frequent expression on melanoma cells9,10,11. HVEM, a member of the tumor necrosis factor (TNF) receptor family, is a transmembrane protein defined structurally by multiple, extracellular, cysteine-rich domains (CRD)1, 2. Intracellularly, HVEM activates NF-κB pathways through interaction with multiple TNF receptor associated factors (TRAFs)12. The overlapping functions of HVEM in immune control result from this combination of complex expression patterns, and the binding of multiple ligands to its extracellular domains.
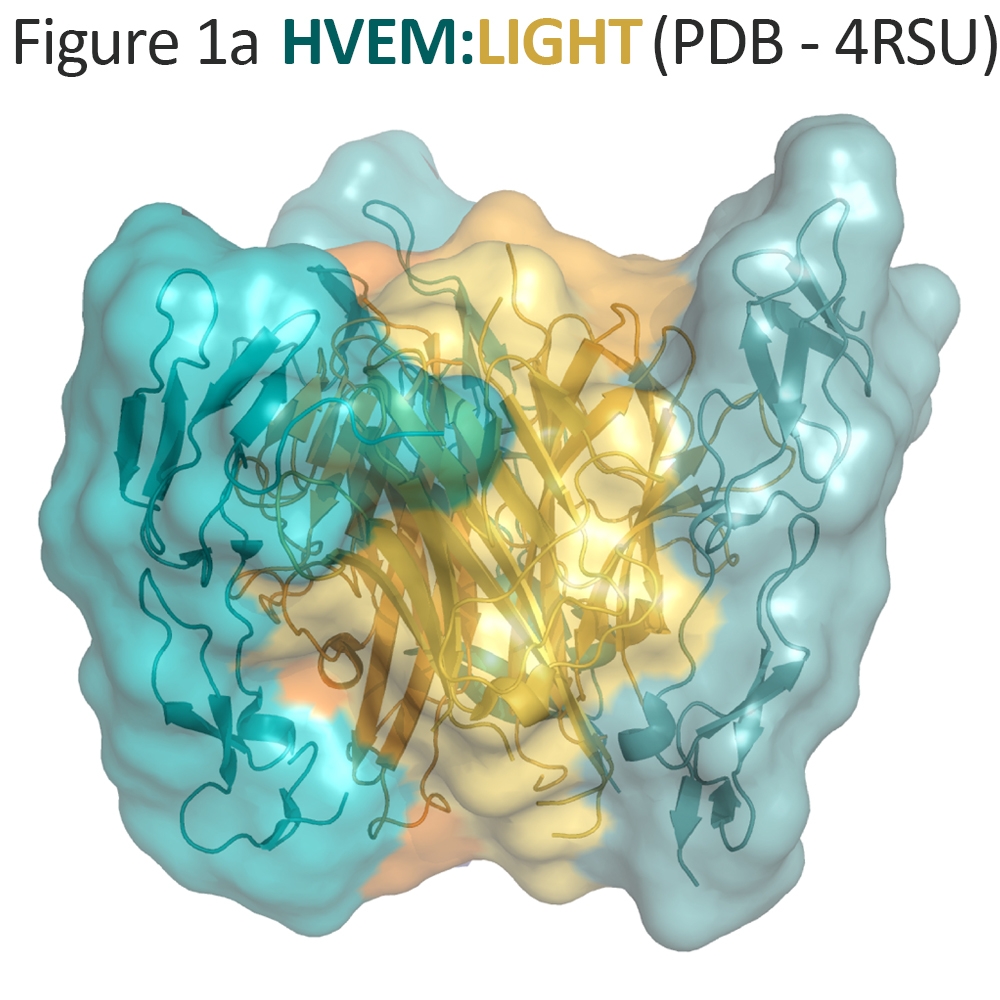
LIGHT is an immune stimulator that contributes to dendritic cell maturation and T-cell expansion4, 13. The protein is expressed by activated T-cells and exists both as a transmembrane protein and a processed, soluble protein14. Both the membrane-bound and soluble configurations of LIGHT can form a trimeric complex that binds directly to the extracellular domain of HVEM15, 16, 17 (Figure 1a). These interactions lead to intracellular signaling from HVEM through the NF-κB pathway stimulating production of pro-inflammatory cytokines and contributing to T-cell growth. Membrane-bound LIGHT has also been implicated as a signaling molecule that operates through increased MAP kinase activity 18, 19. However, LIGHT does not contain an obvious signaling motif and its mechanism for signaling is incompletely defined.
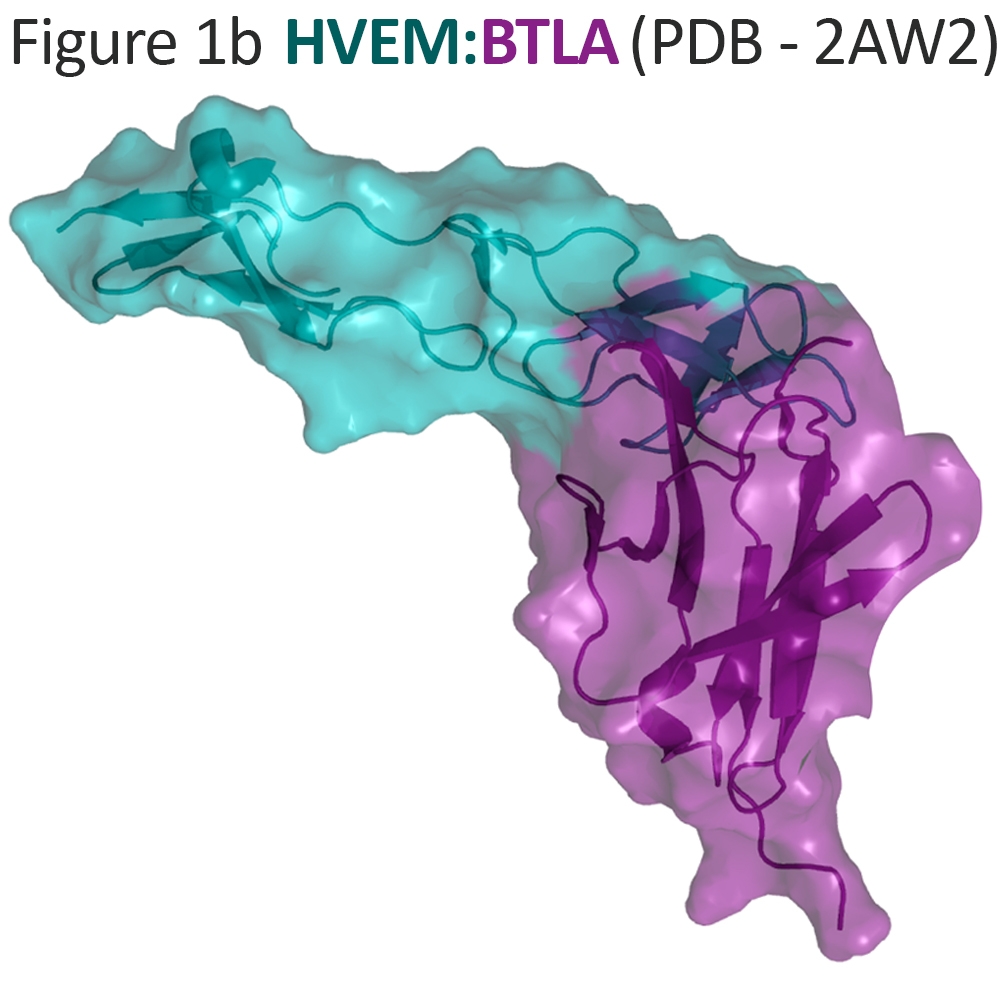
The immune suppressor BTLA functions in opposition to LIGHT in suppression of naïve T-cell expansion7 and induction of Treg cells20. BTLA is an transmembrane protein expressed on B cells, dendritic cells and naive T cells, and activated CD4+ T cells21. The extracellular domain of BTLA binds HVEM through an interface that is distinct and non-competitive with soluble LIGHT17, 22 (Figure 1b).
 Membrane bound LIGHT disrupts this mechanism by a non-competitive mechanism 15. Similar to PD-1 and CTLA4, the intracellular domain of BTLA contains immunoreceptor tyrosine-based inhibition motifs (ITIMs)7, 23, 24. Upon engagement with HVEM, tyrosine phosphorylation within the SH2 domains of the ITMs recruits Shp-1 and Shp-2 resulting of inhibition of NFAT based signaling within the BTLA expressing cell.
Membrane bound LIGHT disrupts this mechanism by a non-competitive mechanism 15. Similar to PD-1 and CTLA4, the intracellular domain of BTLA contains immunoreceptor tyrosine-based inhibition motifs (ITIMs)7, 23, 24. Upon engagement with HVEM, tyrosine phosphorylation within the SH2 domains of the ITMs recruits Shp-1 and Shp-2 resulting of inhibition of NFAT based signaling within the BTLA expressing cell.
CD160 also acts as an immune suppressor through its interactions with HVEM. It is expressed on NK cells and a variety of T cells including CD4+ and CD8+ T cells6. CD160 is a type I membrane protein containing one Ig-V set domain in its extra cellular region25. The extra cellular domain of CD160 binds directly to HVEM through an interaction that is competitive with BTLA17. On this level CD160 and HVEM are somewhat analogous. However, a glycosylphosphatidylinositol anchor connects CD160 to the membrane instead of a transmembrane domain. For this reason, the CD160 mechanism of suppressor signal transduction is not similar to BTLA and PD-1. The exact mechanism of signaling is unclear; however, it is associated with CD3 signaling5.
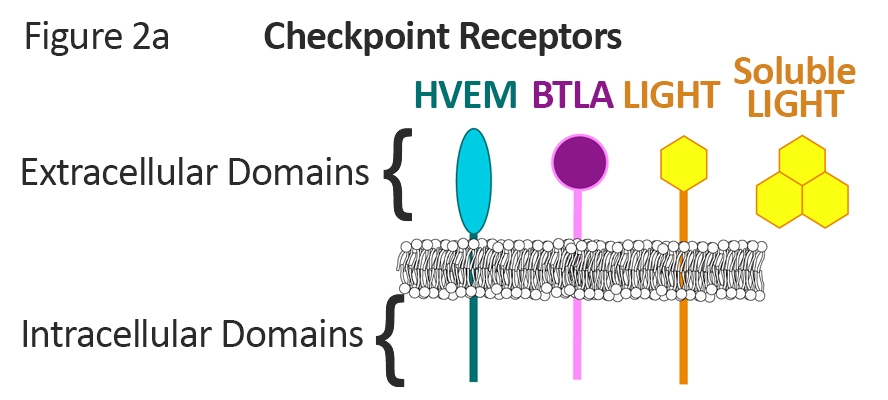
Intricate mechanisms of regulation1, 2 result from the complex patterns of ligand interactions, overlapping expression, and bidirectional signaling (Figure 2a).
Multicomponent complexes lead to varying degrees of inhibitory signaling in cases of cis expression of the checkpoint receptors on the same cell (Figure 2b). In these cases, the default level of immune inhibition occurs through the BTLA-ITAM signaling through the BTLA:HVEM complex. Soluble LIGHT stabilizes this complex forming a BTLA:HVEM:LIGHT complex with a net, immune inhibitory signal. Through a non-competitive binding mechanism15, membrane bound light disrupts the BTLA:HVEM complex. The result is a HVEM:LIGHT complex and alleviation of the BTLA-ITAM signal.
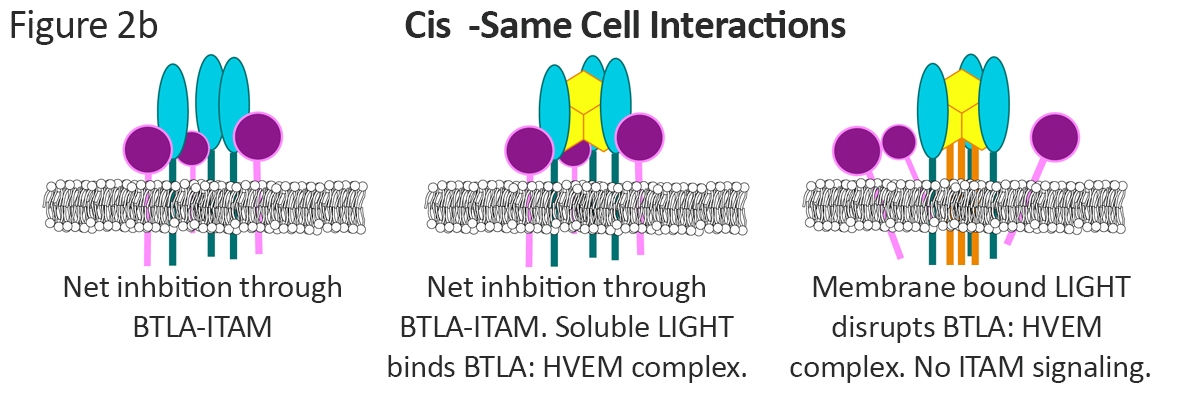
In these cases, the default level of immune inhibition occurs through the BTLA-ITAM signaling through the BTLA:HVEM complex. Soluble LIGHT stabilizes this complex forming a BTLA:HVEM:LIGHT complex with a net, immune inhibitory signal. Through a non-competitive binding mechanism15, membrane bound light disrupts the BTLA:HVEM complex. The result is a HVEM:LIGHT complex and alleviation of the BTLA-ITAM signal.
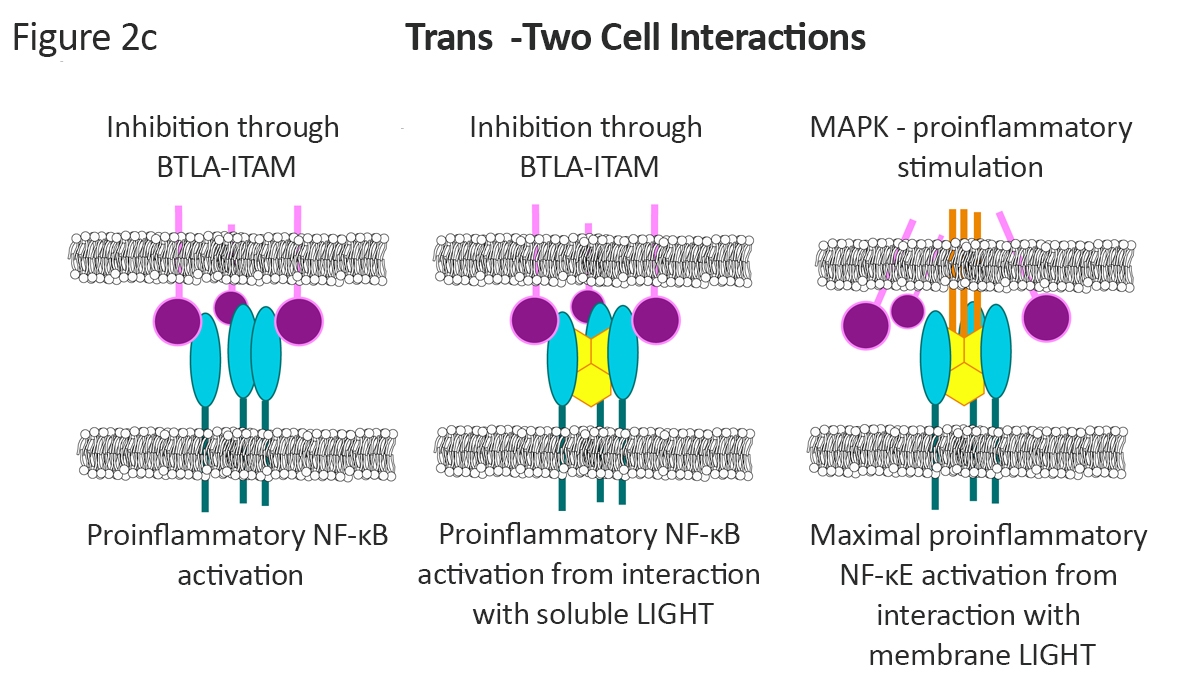
The overlapping signals a tuned to a further degree when the receptors interact through two-cell trans interactions1, 2 (Figure 2c). Here, the BTLA:HVEM complex results in immune inhibitory signals in the BTLA-ITAM expressing cell and proinflammatory NF-kB signaling within the HVEM expressing cell. Soluble LIGHT stabilizes this complex, increasing the signaling within the cells. Finally, membrane bound LIGHT results in a maximal proinflammatory signal by disrupting the HVEM:BTLA complex and forming HVEM:LIGHT. The result is a proinflammatory, NF-kB signal within the HVEM expressing cells, proinflammatory MAPK signaling in the LIGHT expressing cells, and disruption of the BTLA-ITAM signaling.
The immune check point receptors HVEM, LIGHT, CD160, and BTLA are targets for the development of cancer immunotherapies and treatments for immune system related diseases. Success requires relevant, high-quality research tools. BPS Biosciences is a leading provider of HVEM and LIGHT cell lines, proteins, and assay kits.
Cell Lines: HVEM cell lines are available in Jurkat (#79310), HEK293 (#79313), and CHO (#79297) backgrounds. The HVEM NF-κB cell line (#79310) is used in conjunction with the LIGHT-CHO cell line (#79262).
Proteins: BPS provides HVEM proteins as HVEM Fc-fusion (#71142) and as biotinylated HVEM Fc-fusion(#71143). Additionally, BTLA proteins are available as fc-fusion (#71141), fc-fusion-Avi-tagTM (#100033), and biotinylated Fc-fusion, Avi-tagTM with biotin (#100047). LIGHT protiens are offered His-tagged in both human (#71266) and mouse (#79068).
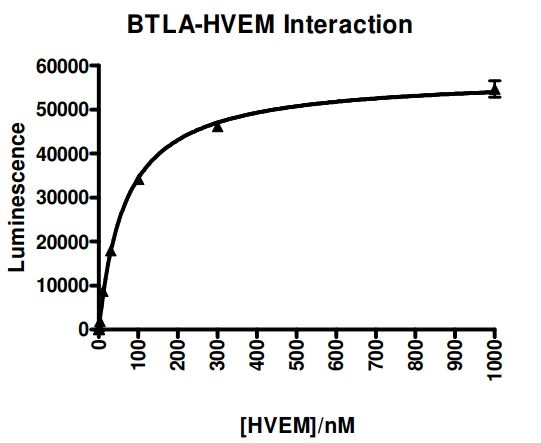
Assay Kit: Using the BTLA:HVEM assay kit (#72008), one can screen for inhibitors of the BTLA: HVEM interaction.
Whatever your research project may entail, BPS Bioscience is here to help you meet your goals. Contact us today to learn more.
References
[1] del Rio, M. L., Schneider, P., Fernandez-Renedo, C., Perez-Simon, J. A., and Rodriguez-Barbosa, J. I. (2013) LIGHT/HVEM/LTbetaR interaction as a target for the modulation of the allogeneic immune response in transplantation, Am J Transplant 13, 541-551.
[2] Murphy, T. L., and Murphy, K. M. (2010) Slow down and survive: Enigmatic immunoregulation by BTLA and HVEM, Annu Rev Immunol 28, 389-411.
[3] Duhen, T., Pasero, C., Mallet, F., Barbarat, B., Olive, D., and Costello, R. T. (2004) LIGHT costimulates CD40 triggering and induces immunoglobulin secretion; a novel key partner in T cell-dependent B cell terminal differentiation, Eur J Immunol 34, 3534-3541.
[4] Morel, Y., Truneh, A., Sweet, R. W., Olive, D., and Costello, R. T. (2001) The TNF superfamily members LIGHT and CD154 (CD40 ligand) costimulate induction of dendritic cell maturation and elicit specific CTL activity, J Immunol 167, 2479-2486.
[5] Cai, G., Anumanthan, A., Brown, J. A., Greenfield, E. A., Zhu, B., and Freeman, G. J. (2008) CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator, Nat Immunol 9, 176-185.
[6] Cai, G., and Freeman, G. J. (2009) The CD160, BTLA, LIGHT/HVEM pathway: a bidirectional switch regulating T-cell activation, Immunol Rev 229, 244-258.
[7] Sedy, J. R., Gavrieli, M., Potter, K. G., Hurchla, M. A., Lindsley, R. C., Hildner, K., Scheu, S., Pfeffer, K., Ware, C. F., Murphy, T. L., and Murphy, K. M. (2005) B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator, Nat Immunol 6, 90-98.
[8] Pasero, C., and Olive, D. (2013) Interfering with coinhibitory molecules: BTLA/HVEM as new targets to enhance anti-tumor immunity, Immunol Lett 151, 71-75.
[9] Derre, L., Rivals, J. P., Jandus, C., Pastor, S., Rimoldi, D., Romero, P., Michielin, O., Olive, D., and Speiser, D. E. (2010) BTLA mediates inhibition of human tumor-specific CD8+ T cells that can be partially reversed by vaccination, J Clin Invest 120, 157-167.
[10] Krieg, C., Han, P., Stone, R., Goularte, O. D., and Kaye, J. (2005) Functional analysis of B and T lymphocyte attenuator engagement on CD4+ and CD8+ T cells, J Immunol 175, 6420-6427.
[11] Otsuki, N., Kamimura, Y., Hashiguchi, M., and Azuma, M. (2006) Expression and function of the B and T lymphocyte attenuator (BTLA/CD272) on human T cells, Biochem Biophys Res Commun 344, 1121-1127.
[12] Schneider, K., Potter, K. G., and Ware, C. F. (2004) Lymphotoxin and LIGHT signaling pathways and target genes, Immunol Rev 202, 49-66.
[13] Zou, G. M., and Hu, W. Y. (2005) LIGHT regulates CD86 expression on dendritic cells through NF-kappaB, but not JNK/AP-1 signal transduction pathway, J Cell Physiol 205, 437-443.
[14] Celik, S., Shankar, V., Richter, A., Hippe, H. J., Akhavanpoor, M., Bea, F., Erbel, C., Urban, S., Blank, N., Wambsganss, N., Katus, H. A., and Dengler, T. J. (2009) Proinflammatory and prothrombotic effects on human vascular endothelial cells of immune-cell-derived LIGHT, Eur J Med Res 14, 147-156.
[15] Cheung, T. C., Steinberg, M. W., Oborne, L. M., Macauley, M. G., Fukuyama, S., Sanjo, H., D'Souza, C., Norris, P. S., Pfeffer, K., Murphy, K. M., Kronenberg, M., Spear, P. G., and Ware, C. F. (2009) Unconventional ligand activation of herpesvirus entry mediator signals cell survival, Proc Natl Acad Sci U S A 106, 6244-6249.
[16] Harrop, J. A., McDonnell, P. C., Brigham-Burke, M., Lyn, S. D., Minton, J., Tan, K. B., Dede, K., Spampanato, J., Silverman, C., Hensley, P., DiPrinzio, R., Emery, J. G., Deen, K., Eichman, C., Chabot-Fletcher, M., Truneh, A., and Young, P. R. (1998) Herpesvirus entry mediator ligand (HVEM-L), a novel ligand for HVEM/TR2, stimulates proliferation of T cells and inhibits HT29 cell growth, J Biol Chem 273, 27548-27556.
[17] Kojima, R., Kajikawa, M., Shiroishi, M., Kuroki, K., and Maenaka, K. (2011) Molecular basis for herpesvirus entry mediator recognition by the human immune inhibitory receptor CD160 and its relationship to the cosignaling molecules BTLA and LIGHT, J Mol Biol 413, 762-772.
[18] Kwon, B. S., Tan, K. B., Ni, J., Oh, K. O., Lee, Z. H., Kim, K. K., Kim, Y. J., Wang, S., Gentz, R., Yu, G. L., Harrop, J., Lyn, S. D., Silverman, C., Porter, T. G., Truneh, A., and Young, P. R. (1997) A newly identified member of the tumor necrosis factor receptor superfamily with a wide tissue distribution and involvement in lymphocyte activation, J Biol Chem 272, 14272-14276.
[19] Shi, G., Luo, H., Wan, X., Salcedo, T. W., Zhang, J., and Wu, J. (2002) Mouse T cells receive costimulatory signals from LIGHT, a TNF family member, Blood 100, 3279-3286.
[20] Jones, A., Bourque, J., Kuehm, L., Opejin, A., Teague, R. M., Gross, C., and Hawiger, D. (2016) Immunomodulatory Functions of BTLA and HVEM Govern Induction of Extrathymic Regulatory T Cells and Tolerance by Dendritic Cells, Immunity 45, 1066-1077.
[21] Murphy, K. M., Nelson, C. A., and Sedy, J. R. (2006) Balancing co-stimulation and inhibition with BTLA and HVEM, Nat Rev Immunol 6, 671-681.
[22] Compaan, D. M., Gonzalez, L. C., Tom, I., Loyet, K. M., Eaton, D., and Hymowitz, S. G. (2005) Attenuating lymphocyte activity: the crystal structure of the BTLA-HVEM complex, J Biol Chem 280, 39553-39561.
[23] Chemnitz, J. M., Lanfranco, A. R., Braunstein, I., and Riley, J. L. (2006) B and T lymphocyte attenuator-mediated signal transduction provides a potent inhibitory signal to primary human CD4 T cells that can be initiated by multiple phosphotyrosine motifs, J Immunol 176, 6603-6614.
[24] Gavrieli, M., Watanabe, N., Loftin, S. K., Murphy, T. L., and Murphy, K. M. (2003) Characterization of phosphotyrosine binding motifs in the cytoplasmic domain of B and T lymphocyte attenuator required for association with protein tyrosine phosphatases SHP-1 and SHP-2, Biochem Biophys Res Commun 312, 1236-1243.
[25] Anumanthan, A., Bensussan, A., Boumsell, L., Christ, A. D., Blumberg, R. S., Voss, S. D., Patel, A. T., Robertson, M. J., Nadler, L. M., and Freeman, G. J. (1998) Cloning of BY55, a novel Ig superfamily member expressed on NK cells, CTL, and intestinal intraepithelial lymphocytes, J Immunol 161, 2780-2790.
Assay Data
BTLA:HVEM binding activity, measured using the using the BTLA:HVEM [Biotinylated] Inhibitor Screening Assay Kit, BPS Bioscience #72008.
HVEM/NF-κB Jurkat Cell Line
The HVEM cell line is a recombinant clonal stable Jurkat T cell line expressing firefly luciferase gene under the control of 4 copies of NF-κB response elements with constitutive expression of human HVEM (#79310).
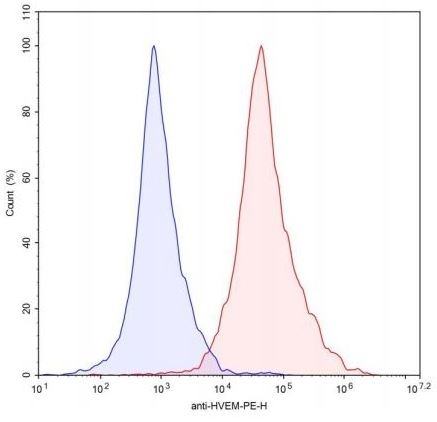
HVEM CHO Cell Line
HVEM-CHO cells (blue) or control CHO cells (red) were stained with PE-labeled AntiHVEM Antibody (Biolegend #318806) and analyzed by FACS. (#79297)
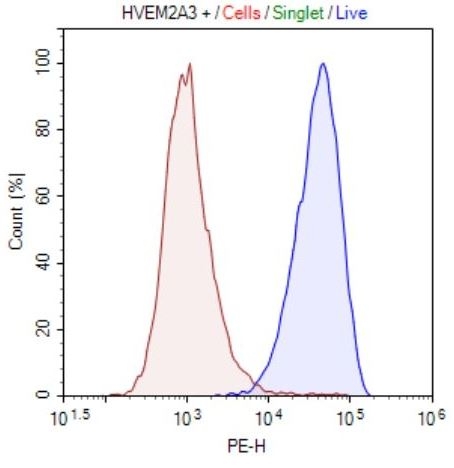
HVEM HEK293 Cell Line
HVEM-HEK293 cells (red) or control HEK293 cells (blue) were stained with PE-labeled Anti-HVEM Antibody (Biolegend, #318806) and analyzed by FACS. (#79313)
LIGHT CHO Cell Line
Flow cytometry showed APC-conjugated anti-human LIGHT antibody (Abcam, #ab155363) detects LIGHT-positive clonal population (clone A4) (blue) (#79262), using wild-type CHO cells as a negative control (red).
Proteins
Cell Lines
Assay Kits
Services