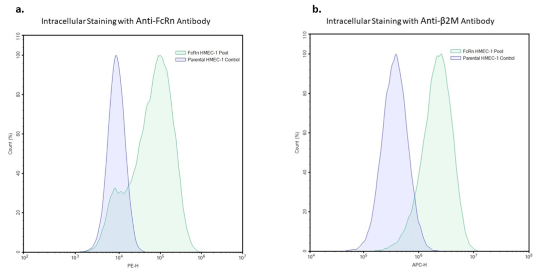
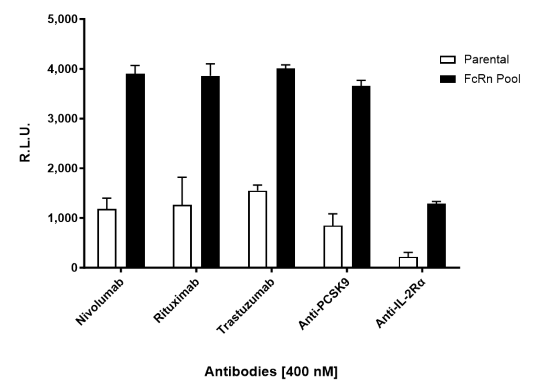
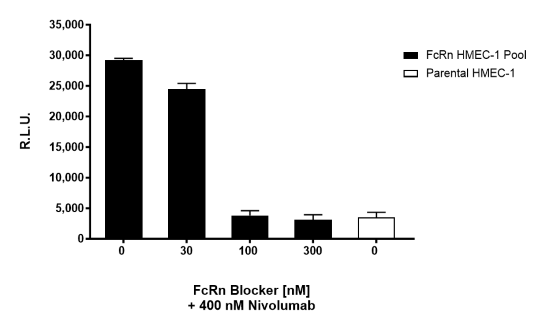
FcRn: IgG Recycling HMEC-1 Cell Pool
The FcRn: IgG Recycling HMEC-1 Cell Pool is a HMEC-1 cell pool expressing Fc gamma receptor and transporter (FCGRT) (NM_004107.5) and beta-2-microglobulin (B2M) (NM_004048.4), separated by a T2A self-cleaving peptide, under the control of a CMV promoter, introduced into HMEC-1 cells via lentiviral transduction. This cell pool is designed to measure the uptake and recycling of human IgG antibodies (human FcRn does not bind mouse IgG).
This cell pool has been validated with Nivolumab, Rituximab, Trastuzumab, anti-PCSK9 and an anti-IL-2Ra antibody.
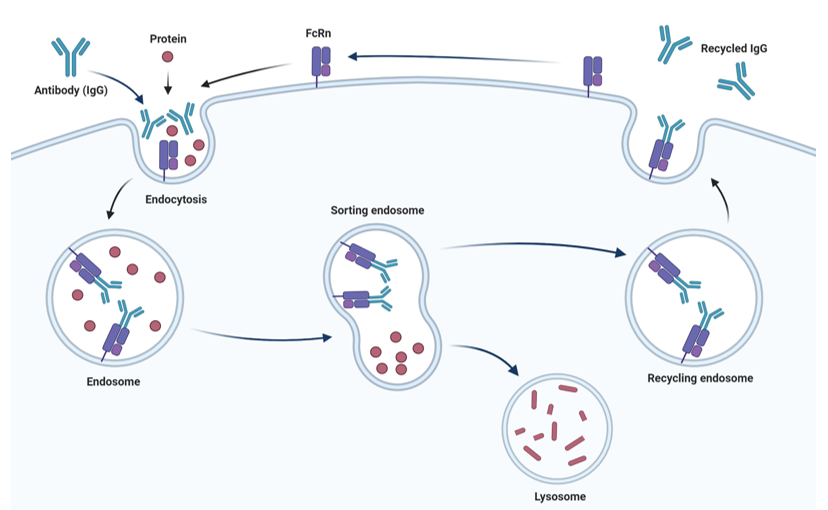
Figure 1: Illustration of FcRn: IgG Recycling HMEC-1 Cell Pool mechanism of IgG FcRn-mediated recycling.
Cells are incubated with an antibody (IgG) of interest in a low pH Dilution Buffer, allowing FcRn to bind to IgG. Cells are then washed, and IgG recycling can be analyzed after 24 hours. The recycled antibody will be present in the cell culture media. If the antibody is retained inside the cells, detection can be done by lysing the cells and measuring the cellular content by ELISA or another method of interest.
Interested in profiling FcRn-mediated antibody update and recycling or test FcRn inhibitors without the need to purchase and license the cell line? Check out our Immunotherapy Cell-Based Screening Services.
Purchase of this cell line is for research purposes only; commercial use requires a separate license. View the full terms and conditions.
Media and Supplements Required for Cell Culture
| Name | Ordering Information |
| Thaw Medium 18 | BPS Bioscience #82205 |
| Growth Medium 18A | BPS Bioscience #82206 |
| Assay Medium 8A | BPS Bioscience #82207 |
| EGF Recombinant | BPS Bioscience #90201-3 |
Materials Used in the Cellular Assay
| Name | Ordering Information |
| FcRn Recycling Wash Buffer (pH 7-7.4) | BPS Bioscience #82208 |
| FcRn Recycling Dilution Buffer (pH 6) | BPS Bioscience #82209 |
| Human IgG ELISA Kit | Novus #NBP3-00400 |
| Nivolumab (Anti-PD-1 Antibody) | Selleckchem #A2002 |
| Rituximab (Anti-CD20 Antibody) | Selleckchem #A2009 |
| Trastuzumab (Anti-HER2 Antibody) | Selleckchem #A2007 |
| Human Proprotein Convertase 9/PCSK9 Antibody | R&D Systems #MAB10347 |
| Human CD25/IL-2Rα Antibody | R&D Systems #MAB9926 |
| FcRn (FCGRT/B2M) Blocker | BPS Bioscience #101468 |
| EGF Recombinant | BPS Bioscience #90201-3 |
| PD-1, FLAG-Avi-His-Tag, Biotin-Labeled (Human) Recombinant | BPS Bioscience #71325 |
| 96 well plates | |
| Plate reader compatible with ELISA |
The cell line has been screened to confirm the absence of Mycoplasma species.
Neonatal Fc receptor for IgG (FcRn) is a heterodimeric protein. FcRn consists of Fc Gamma Receptor and Transporter, encoded by the FCGRT gene, and beta-2-Microglobulin (B2M). FcRn binds to the Fc region of monomeric immunoglobulin G (IgG). It is expressed in over 25 tissue types, with high expression levels observed in the spleen and intestine. In the placenta, it transports IgGs from the mother to the fetus. FcRn contributes to effective humoral immunity by protecting IgGs from degradation, recycling them and extending their half-life in circulation. In addition to IgGs, it regulates the homeostasis of serum albumin. FcRn plays a vital role in regulating the level of albumin and IgGs in circulation by binding albumin and the Fc region of IgG at low pH (about pH 6.0) in endosomes. It then diverts them from being degraded by lysosomes and instead recycles them for release into the neutral pH (about 7.0-7.4) of the extracellular compartment. The loss of FcRn in tumor cells has been reported to play a role in cancer by increasing the breakdown of albumin into amino acids needed for tumor cells proliferation. The function of FcRn can be exploited by engineering therapeutic antibodies to increase their binding to FcRn, thereby improving their half-life and therapeutic efficacy. For example, an antibody cocktail that contains Fc mutations, and thus an extended half-life (Evusheld) has been used to treat COVID-19. There are now several other drugs in clinical using similar strategies. Further studies and drug development taking advance of FcRn properties will provide new therapeutic options.
Grevys A, et al., 2018 Nature Communications, 9: 621.
Swiercz R, et al., 2017 Oncotarget, 8(2): 3528–3541.
Weflen A, et al., 2013 Mol Biol Cell, 24(15): 2398–2405.