FcRn (FCGRT/B2M) Blocker
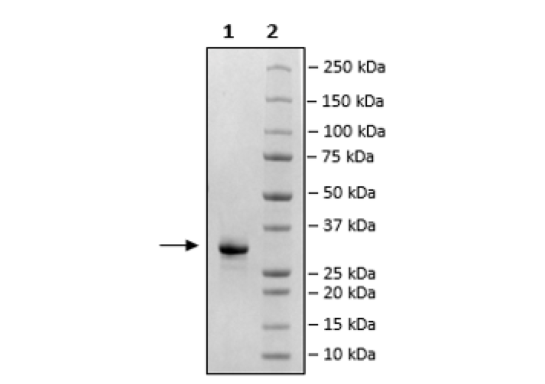
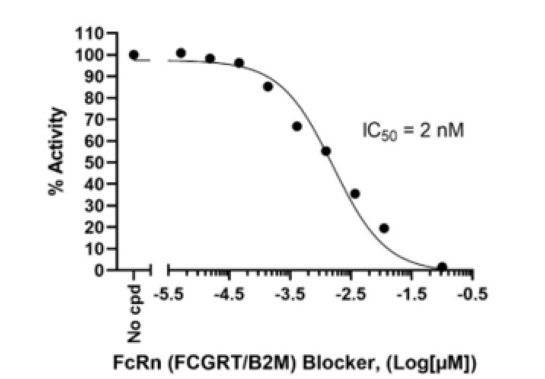
The FcRn (FCGRT/B2M) Blocker (Fc-Tag) is a recombinant human protein corresponding to the Fc region of an antibody which has been engineered to bind to the human neonatal Fc Receptor of IgG (FcRn; FCGRT/B2M) with high affinity. This protein has been tested for specific binding to human FcRn (BPS Bioscience #71283) and it blocks the binding of FcRn to IgG. The FcRn blocker is comparable to Efgartigimod alfa-fcab. This protein was affinity purified.
≥90%
Aqueous buffer solution
8 mM phosphate, pH 7.4, 110 mM NaCl, 2.2 mM KCl, and 20% glycerol
Neonatal Fc receptor for IgG (FcRn) is a heterodimeric protein consisting of the Fc Gamma Receptor and Transporter encoded by the FCGRT gene, associated with beta-2-Microglobulin (B2M). FcRn binds to the Fc region of monomeric immunoglobulin G (IgG). FcRn contributes to an effective humoral immunity by protecting IgGs from degradation, recycling them and extending their half-life in circulation. In addition to IgGs, it regulates the homeostasis of serum albumin. The loss of FcRn in tumor cells has been reported to play a role in cancer by increasing the breakdown of albumin to amino acids needed by tumor cells for proliferation. The function of FcRn can be exploited by engineering therapeutic antibodies to increase their binding to FcRn, thereby improving their half-life and therapeutic efficacy. For example, an antibody cocktail that contains Fc mutations and an extended half-life (Evusheld) is used to treat COVID-19. There are now several other drugs in clinical using similar strategies.