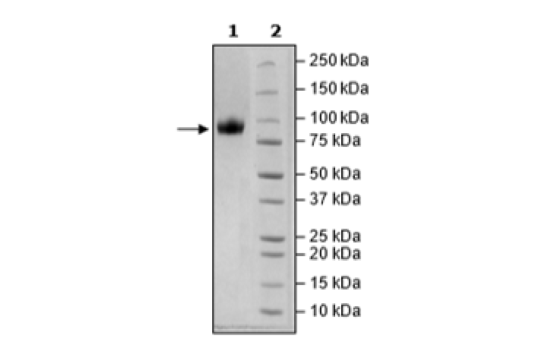
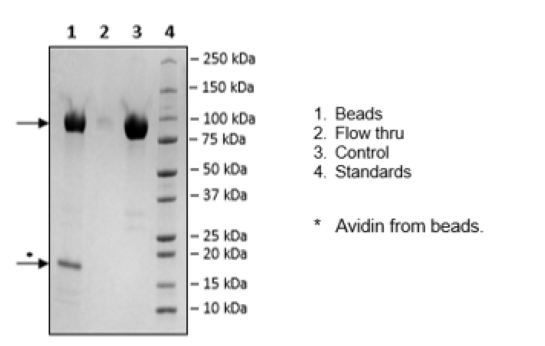
CD155, Fc Fusion, Avi-Tag, Biotin-Labeled Recombinant
Recombinant human CD155, encompassing amino acids 27-343. This construct contains a C-terminal human IgG1 Fc domain followed by an Avi-Tag™. This protein was affinity purified.
≥90%
Aqueous buffer solution.
8 mM phosphate, pH 7.4, 110 mM NaCl, 2.2 mM KCl, and 20% glycerol
This protein is enzymatically biotinylated using Avi-Tag™ technology. Biotinylation is confirmed to be ≥90%.
For more information on enzymatic biotinylation, please see our Tech Note.
CD155 (also known as PVR cell adhesion molecule) is a transmembrane glycoprotein receptor of the immunoglobulin superfamily of proteins. It is highly expressed in dendritic cells, fibroblasts, endothelial cells, and multiple tumor cells including ovarian carcinoma, non-small cell lung, glioblastoma, and colorectal carcinoma. It has a high affinity for a T cell regulatory transmembrane surface protein called TIGIT (T cell immunoglobulin and ITIM domains) and plays a key role in tumor cell invasion and migration. Interaction between CD155 and TIGIT causes immunosuppressive effects on CD4+ and CD8+ T cells, which hinders T cell proliferation and function. CD155 also interacts with the regulatory receptors CD226 (expressed on natural killer (NK) cells, monocytes and CD4+ T cells) and CD96. While CD155-CD226 engagement activates NK cell cytotoxicity and T cell response, CD155-CD96 interaction inhibits NK cell function. Due to these opposing regulatory effects, blockade of CD155-TIGIT and CD155- CD226 interactions are attractive therapeutic strategies in cancer immunotherapy and autoimmune diseases, respectively.