SiMPLe CHC Antibody Labeling Kit (Sortase Mediated Protein Ligation)
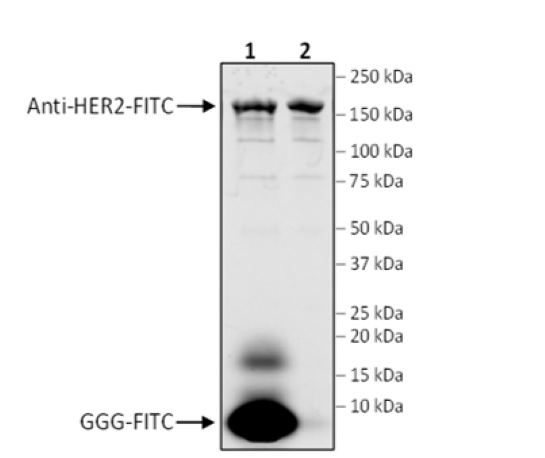
The SiMPLe CHC Antibody Labeling Kit leverages Sortase Mediated Protein Ligation to specifically label recombinant antibodies containing the sequence LPXTG at the C-terminus of the heavy chain (CHC). A diverse number of poly-Glycine (G)n molecules and labels (e.g. fluorophores, biotin, enzymes, peptides, etc.) are compatible with this kit. The site-specific conjugation ensures that the antigen binding site remains available for target binding and reduces heterogeneity compared to chemical conjugation methods, making it ideal for Antibody-Drug Conjugate (ADC) development and flow cytometry. The kit provides reagents to label 3 x 100 μg of recombinant antibody, as well as purification columns to aid in the removal of excess poly-Glycine (G)n-label. Anti-HER2-LPETGH6 and GGGK-FITC are included as positive controls.
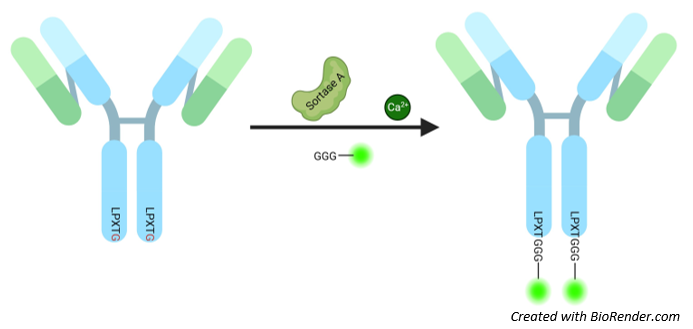
Figure 1: Illustration of the mechanism of targeted labeling using the SiMPLe CHC Antibody Labeling Kit (Sortase Mediated Protein Ligation).
Each antibody contains two Sortase recognition sequences, so a maximum of two labels per antibody can be achieved
- Antibody of interest containing the sequence LPXTG in the C-terminus of the heavy chain (1 mg/ml, 100 μg per reaction)
- Poly-Glycine-Label (125 μM per reaction, 3.75 mM stock)
- Microcentrifuge
- Buffer for final formulation (such as PBS or TBS)
| Catalog # | Name | Amount | Storage |
| 71048 | Sortase A Heptamutant, His-Tag | 3 Vials (1U/Vial) | -80°C |
| 79394 | Reaction Buffer | 100 μl | 4°C |
| 82197 | Stop Solution | 20 μl | 4°C |
| 79396 | Purification Columns | 3 | Room Temp |
| 79397 | Collection Tubes | 6 | Room Temp |
| 101689 | Anti-HER2-LPETGH6 | 1 Vial | -80°C |
| 82198 | GGG-FITC | 1 Vial | -80°C |
Staphylococcal Sortase A is a bacterial transpeptidase that covalently attaches proteins to the bacterial cell wall, maintaining bacterial virulence and infectivity. Sortase A cleaves a specific peptide sequence (LPXTG recognition motif) within a target protein between threonine and glycine, with a strong preference for terminal locations in proteins. The cysteine residue of the active site forms a transient thioacyl intermediate complex with the substrate protein. This intermediate complex is then immediately attacked by oligo-glycine nucleophiles present on peptide-glycans of the bacterial wall to form an amide bond. This transpeptidase activity can be used for protein labeling. Using a recombinantly expressed antibody containing a C-terminal Sortase recognition sequence (LPXTG for S. aureus Sortase A) and a highly active Sortase A Heptamutant, the direct conjugation of poly-Glycine (G)n-labels to the target protein can be achieved.
Popp M., 2015 Methods Mol Biol. 1266:185-98.