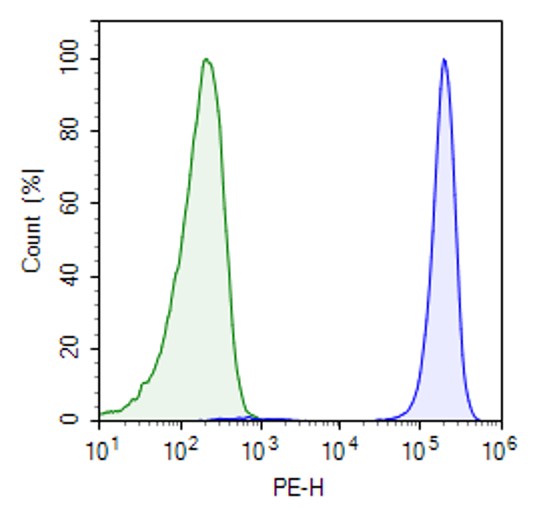
LILRB4 NFAT-Luciferase Reporter Jurkat Recombinant Cell Line
LILRB4 NFAT-Luciferase Reporter Jurkat Cell Line are Jurkat cells stably expressing human LILRB4 (Leukocyte Immunoglobulin-Like Receptor Subfamily B Member 4, GenBank Accession #NM_001278426) and firefly luciferase under the control of NFAT response elements. Activation of NFAT in these cells can be monitored by measuring luciferase activity.
This cell line was validated by flow cytometry for expression of LILRB4.
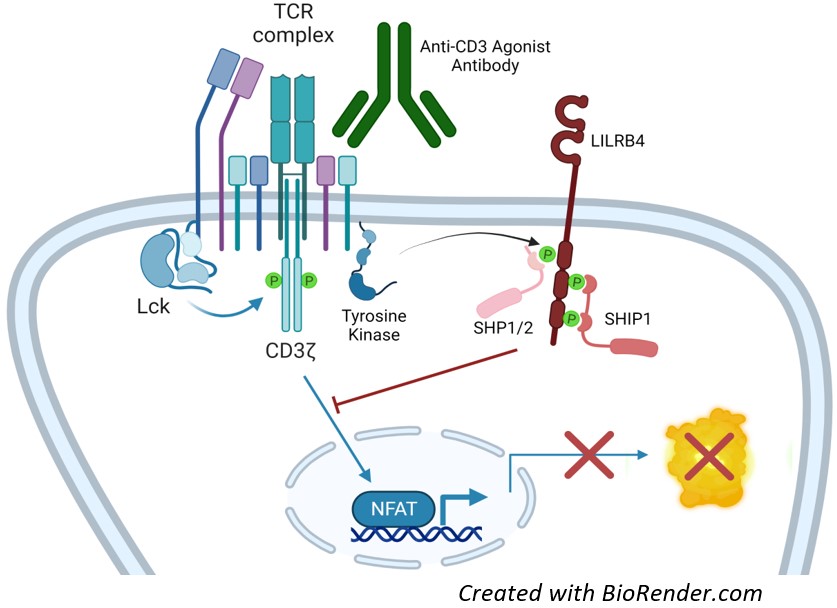
Figure 1: Mechanism of action of LILRB4 NFAT Luciferase Reporter Jurkat Cell Line.
In response to TCR activation, LILRB4 is tyrosine phosphorylated by Src-family kinases such as Lck or Fyn, which triggers recruitment of SH2 domain-containing phosphatases, leading to attenuation of the T cell activation signals
Interested in screening and profiling inhibitors, blocking antibodies, or activators of LILRB4 without the need to purchase and license the cell line? Check out our Cell Signaling Pathway Screening.
Purchase of this cell line is for research purposes only; commercial use requires a separate license. View the full terms and conditions.
| Name | Ordering Information |
| Thaw Medium 2 | BPS Bioscience #60184 |
| Growth Medium 2F | BPS Bioscience #79669 |
The cell line has been screened to confirm the absence of Mycoplasma species.
Leukocyte Immunoglobulin-Like Receptor Subfamily B Member 4 (LILRB4, also known as B4, ILT3, LIR5, or CD85K) is a transmembrane immune checkpoint found in leukocytes (for example macrophages and dendritic cells) and involved in adaptive immunity. It binds MHC class I molecules at the surface of antigen-presenting cells, plays a role in antigen presentation, and transduces an inhibitory signal meant to down-regulate immune responses. Thus, it inhibits T cell proliferation, induces anergy, and regulates the development of immune tolerance.
The protein contains two extracellular immunoglobulin-like domains, a transmembrane domain, and three cytoplasmic immunoreceptor tyrosine-based inhibitory motifs (ITIMs) that allow it to be phosphorylated by cytosolic tyrosine kinases. Once phosphorylated, the ITIMs bind to SH2-domain containing phosphatases SHP-1/2 (SH2 domain-containing phosphatase 1 and 2) and SHIP-1 (Src homology 2 (SH2) domain containing inositol polyphosphate 5-phosphatase 1). SHP-1 dephosphorylates a wide spectrum of phospho-proteins involved in hematopoietic cell signaling whereas SHIP-1 dephosphorylates lipids at the plasma membrane and serves as a docking molecule. Both phosphatases are negative regulators of immune cell proliferation.
Differential LILRB4 expression levels have been observed in immune diseases such as Kawasaki disease, systemic lupus, and sepsis. Expression of the protein in Acute Myeloid Leukemia (AML) cells suppresses anti-tumor T cell activity. Because LILRB4 is highly expressed on AML cells but not on normal hematopoietic stem cells, it is considered a promising therapeutic target for CAR-T cells. It is also an immunotherapy target due to its role as an immune suppressive checkpoint. Indeed, blocking LILRB4 function is expected to improve anti-tumor immunity or to decrease auto-immunity and pathological inflammation.
Deng M., et al., 2018, Nature 562(7728): 605-609.
John S., et al., 2018 Mol Ther.26(10): 2487-2495