ARG1/ARG2 Screening Services
Arginase enzymes convert arginine to ornithine through hydrolysis. Two known isoforms of Arginase exist, ARG1 and ARG2. This enzyme is involved in the regulation of a variety of immunological responses and is a major target in immunotherapy. ARG1/2 is overexpressed in myeloid-derived suppressor cells (MDSCs) and tumor-associated macrophages (TAMs). Overexpression of ARG1/2 results in depleted levels of arginine both intracellularly and extracellularly. As arginine levels are depleted in the microenvironment, immune cells are starved of this amino acid and the function of key immunological activators become impaired; T cell proliferation is inhibited, regulatory T cells become activated and inhibit CD4+ T cells, and immunosuppressants have increased longevity. Depleted arginine also results in the release of reactive nitrogen species and reactive oxygen species from TAMs and MDSCs. These reactive species cause T cell apoptosis and the activation and growth of antigen presenting cells.
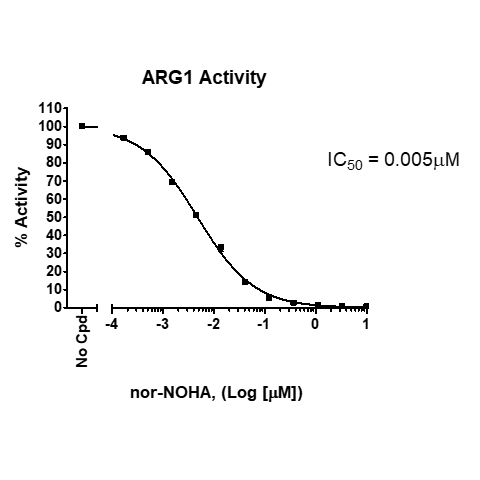
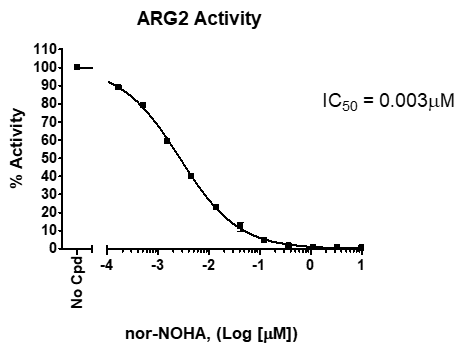
Due to their essential regulation of immune function, ARG1/2 are an important drug targets in immunotherapy. In addition to the ARG1 and ARG2 proteins, BPS offers ARG1/2 screening services. Using BPS’s ARG1/2 screening services, we can help accelerate your research and drug discovery programs using our arginase bioassays.
Trust us to provide you with high quality data with fast turnaround time. Our team of experts along with our broad services portfolio makes it easy to:
- Screen for inhibitors/targets using our ARG assay
- Select from IC50 determination and single point concentrations
- Receive data within days of compound submission
- Perform follow-up studies using the same soluble receptors manufactured in-house
- Get questions anwered or project guidance in a time-efficient manner