IDO/TDO Pathway
BPS Bioscience is a trusted provider and manufacturer of IDO pathway reagents, assay kits, cell lines and services. Cancer cells use the indoleamine 2,3-dioxygenase (IDO) and tryptophan 2,3-dioxygenase (TDO) pathways to suppress the host’s immune response in order to facilitate survival, growth, invasion, and metastasis of malignant cells. IDO enzymes and IDO-related enzymes are active in many tumors, providing a direct defense against T cell attack. IDO is also active in many antigen-presenting cells, resulting in peripheral tolerance to tumor-associated antigens. Key enzymes involved in this and related pathways include IDO1 enzyme, IDO2 enzyme, TDO enzyme, tryptophan hydroxylase 1 and 2 (TPH1 enzyme and TPH2 enzyme).
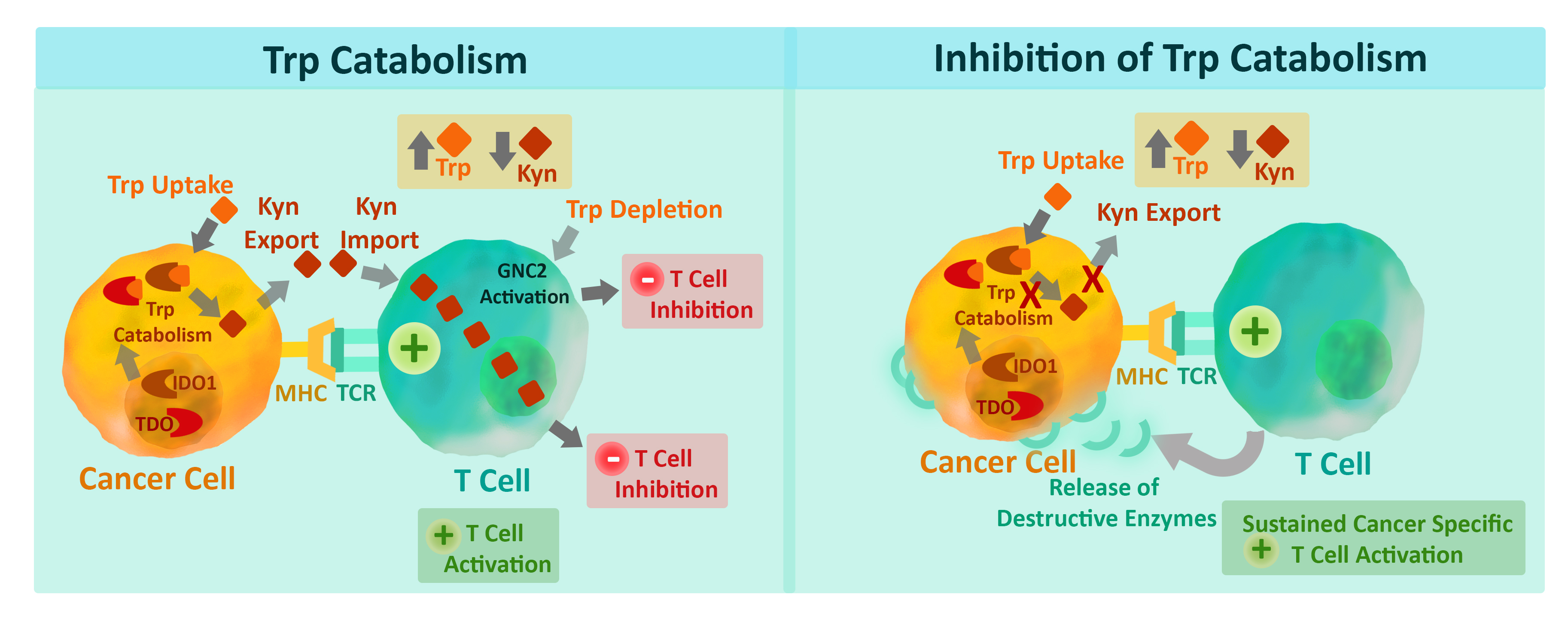
The mechanism of IDO and TDO suppression of tumor immunity involves both tryptophan (Trp) depletion and synthesis of immunosuppressive metabolites, such as kynurenine (Kyn). Trp is an important and essential amino acid that has a variety of functions within the cell. Cancer cell-expressed IDO1, IDO2, or TDO catalyzes the first and rate limiting step of the Kyn-Trp degradation pathway. The two IDO isozymes, IDO1 enzyme and IDO2 enzyme, are normally involved in the metabolic breakdown of the amino acid tryptophan. IDO enzyme expression also directly activates the regulatory T cells that shut down the immune response, which amplifies the suppressive effect.
Shown below is the role of the IDO1 enzyme and TDO enzyme in cancer cell immune evasion. IDO1 enzyme and TDO enzyme catalyze the conversion of Trp to Kyn. Depletion of Trp and increased levels of Kyn in the tumor microenvironment combine to produce an inhibitory signal in T cells that prevents them from attacking the cancer cell. Inhibition of IDO1 enzyme and/or TDO-catalyzed Trp catabolism relieves the immunoinhibitory microenvironment.
Immune cells that are largely responsible for tumor immunity, cytotoxic T cells (CTL) and natural killer (NK) cells are acutely sensitive to Trp depletion, and lowered Trp levels leads to decreased CTL and NK cell proliferation and activation. Additionally, the Trp metabolite, Kyn, can induce T cell differentiation into FoxP3+ regulatory T cells, which further suppresses the host immune system. All of this facilitates survival, growth, invasion, and metastasis of malignant cells 4 (Fig. 1). IDO pathway inhibitors, including NLG919 and INCB02460 can prevent this immunosuppression and allow the activation of immune cells that recognize and fight the cancer cells in pre-clinical models 5,6. A number of IDO and TDO inhibitors have now advanced to clinical trials.
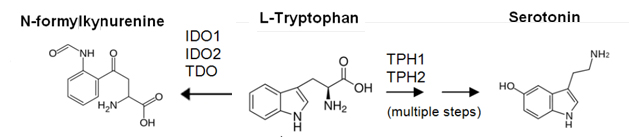
Despite their promise, it remains unclear what off-target effects IDO and TDO inhibitors may have, as Trp is not only catabolized by IDO and TDO. Two other important enzymes, TPH1 enzyme and TPH2 enzyme, are involved in Trp catabolism via a pathway that ultimately leads to the synthesis of serotonin and dopamine, two important neurotransmitters. Given that IDO, TDO, TPH1, and TPH2 enzymes share Trp as a substrate, it may be wise for drug developers to test for IDO/TDO inhibitor cross-reactivity against TPH1/2. Indeed, there is preclinical evidence that suggest inhibiting TPH enzymes can also relieve immunosuppression.
Shown here is a depiction of two important tryptophan catabolic pathways and the enzymes that participate in the process. IDO1, IDO2, and/or TDO inhibitors may exhibit off target effects on TPH enzymes (TPH1 enzyme & TPH2 enzyme). Additionally, IDO/TDO pathway inhibitors may drive tryptophan catabolism via the TPH pathway.
Substantial research efforts have focused on the identification and development of inhibitors of the various Trp catabolic enzymes.
Screen for IDO or TDO inhibitors in BPS Bioscience's IDO1 stable cell line, IDO2 stable cell line, TDO stable cell line and IDO and TDO cell-based assay kits. These kits are easy to use and provide researchers a tool to screen for IDO and TDO inhibitors in a cellular context
If you need assistance running your screens, check out our Immunotherapy Screening Services. In addition to IDO / TDO cell based assay kits, BPS is a provider of IDO1, IDO2 and TDO inhibitor screening assay kits and related buffers and supplements. BPS Bioscience’s Immunotherapy Screening Services allow you to screen for inhibitors of any of many targets in our biochemical assays including IDO1, IDO2 and TDO.
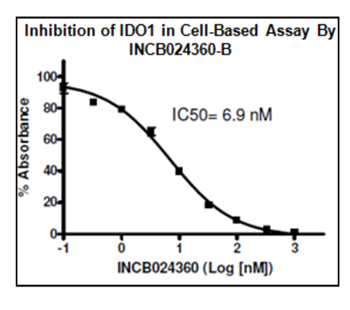
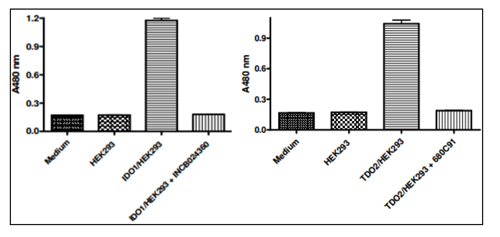
Inhibition of IDO1 activity by BPS’ IDO1 Cell-Based Assay Kit (BPS Cat. #72031) using INCB024360-B demonstrating BPS’ cell-based screening and profiling services.
References:
1. Platten, M., et al., “Tryptophan catabolism in cancer: beyond IDO and tryptophan depletion.” Canc. Res. 72.21 (2012):5435-5440.
2. Platten, M., et al., “Cancer Immunotherapy by Targeting IDO1/TDO and Their Downstream Effectors.” Front. Immunol. 5.673 (2015).
3. Stavrum, A-K., et al., “Model of Tryptophan Metabolism, Readily Scalable Using Tissue-specific Gene Expression Data.” J. Biol. Chem. 288.48 (2013). 34555-34566.
4. Munn, D.H., and Mellor, A.L. “Indoleamine 2,3-dioxygenase and tumor-induced tolerance.” J. Clin. Invest. 117 (2007):1147-1154.
5. Liu, X. et al., “Selective inhibition of IDO1 effectively regulates mediators of antitumor immunity.” Blood. 115.17 (2010):3520-3530.
6.Pilotte, L. et al., “Reversal of tumoral immune resistance by inhibition of tryptophan 2,3-dioxygenase.” Proc. Nat. Acad. Sci USA. 109.7 (2012):2497-2502.
7.Nowak, E.C., et al., “Tryptophan hydroxylase-1 regulates immune tolerance and inflammation.” J. Exp. Med. 209.11. (2012):2127-2135.
Block and mediate Immune Checkpoints (Inhibitors & Controls)
Take advantage of our IDO inhibitors and controls. Manipulate and intermediate the TRP and TPH pathways.
Detect IDO Enzyme and TDO Enzyme Expression
Monitor IDO and TDO expression with qualified Inhibitor Screening Assay Kits. Two kit formats allow more detection methods for extra validation, Homogenous and Fluorogenic.
Monitor IDO and TDO T-Cell Responses
Explore reactions on a cellular context utilizing BPS' IDO and TDO cell based assay kits.
Screen for inhibitors of IDO and TDO
Dive deeper in the cellular context level with easy to use Stable Cell Lines.
Custom Solutions
Let us fuel your research. Partner up with our product development scientists and attain reagents specific to your needs.
Screening and Profiling Solutions
Fast turnaround services for compound screening, toxicity measurements and proliferation analysis
Connect With BPS
Request a Quote Send Compounds Contact us