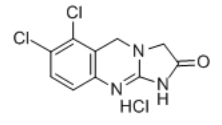
Anagrelide HCl
Catalog #
27732-1
$120
*
●
●
Purchase
Description
Anagrelide is a selective thrombocytopenic agent that inhibits PDE3. It also has been used in the treatment of chronic myeloid leukemia.
●
Synonyms
6,7-dichloro-5,10-dihydro-3H-imidazo[2,1-b]quinazolin-2-one;hydrochloride
●
Product Data Gallery
Product Info
Storage and Usage
Citations
Target(s)
PDE3
Formula
C10H7Cl2N3O • HCl
MW
292.6 Da
Solubility
Soluble in DMSO (10 mM), water (<1 mg/ml)
Biological Activity
The exact mechanism by which anagrelide reduces blood platelet count is unclear, although it is known to be a potent inhibitor of PDE3 (IC50 = 36nM).
CAS Registry #
58579-51-4
Background
Anagrelide is a FDA-approved drug for the treatment of essential thrombocythemia. It is originally identified as a potential inhibitor of platelet aggregation. Anagrelide selectively affects thrombocytes while shows no significant effect on white blood cells, erythrocytes, or coagulation. Anagrelide is a potent inotropic agent for dogs with remarkable vasodilatory activity. Additionally, anagrelide can reduce renal blood flow. Furthermore, anagrelide has shown to play roles in other chronic myeloproliferative disorders, such as polycythemia vera, chronic myeloid leukemia and agnogenic myeloid metaplasia. Anagrelide has some side effects involving headache, diarrhea, edema, palpitations, and abdominal pain.
References
Oertel MD. Am J Health Syst Pharm. 1998 Oct 1;55(19):1979-86.